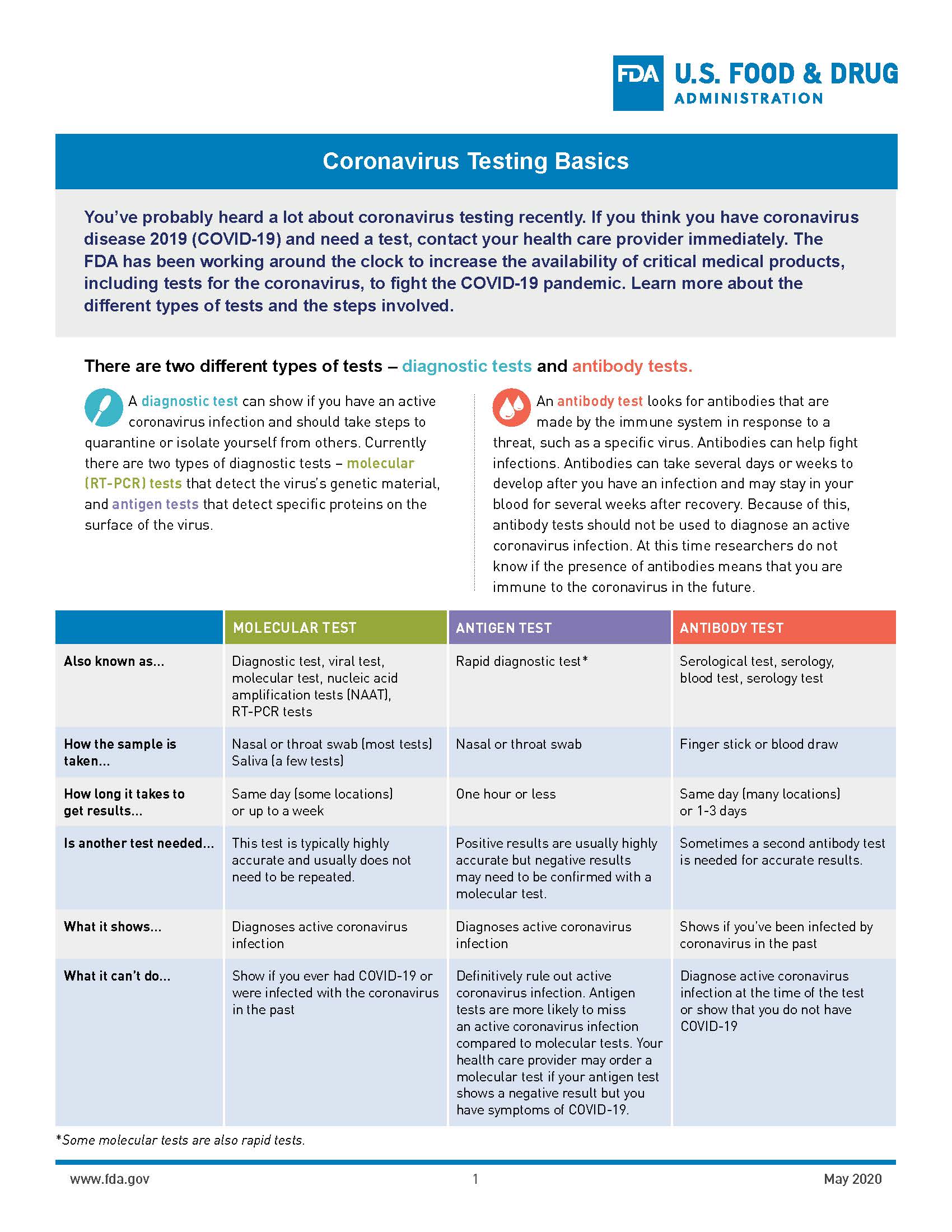
COVID-19 Testing
Testing
If you think you have been exposed to COVID-19 and develop a fever and symptoms, such as cough or difficulty breathing, call your healthcare provider for medical advice. Please do not show up without calling first. Clinicians will use their judgment to determine if a patient has signs and symptoms compatible with COVID-19 and whether the patient should be tested.
Effective February 5, 2020, the “2019 Novel Coronavirus” was added to the Reportable Diseases, Emergency Illnesses and Health Conditions, and Reportable Laboratory Findings list.
CT DPH is requesting that physicians use the Electronic 2019 Novel Coronavirus (COVID-19) Case Report Form.
Drive-Up Testing Sites
In addition to the State Department of Public Health Laboratory, healthcare systems across Connecticut are partnering with private clinical laboratories, like Quest Diagnostics and LabCorp to conduct COVID-19 testing. These private labs have been issued Emergency Use Authorization from the Food and Drug Administration (FDA) to conduct COVID-19 diagnostic testing. As of now, the testing results take approximately 4 days to receive. Patients must have a referral from a provider and call ahead to make an appointment.
Local Locations (for additional sites in CT, please call 2-1-1):
- Griffin Hospital
- Call the Griffin Health Hotline at 203-204-1053. Griffin Health is offering a call center for patients, visitors and members of the community who have questions about COVID-19. Griffin Health caregivers are available to answer your specific questions Monday – Friday from 8 a.m. – 5 p.m.
- Yale-New Haven Hospital
- YNHHS COVID-19 Call Center at 833-ASK-YNHH (833-275-9644)
- St. Mary’s Hospital
- Waterbury Hospital
- Bridgeport Hospital
- Some CVS locations in CT are now offering drive-up COVID-19 testing.
- Click here for more information and requirements: https://www.cvs.com/minuteclinic/covid-19-testing
CDC Fact Sheet for Patients Tested for COVID-19
On February 28, 2020 Governor Lamont Announced Connecticut’s Public Health Lab Approved to Run Diagnostic Testing for Coronavirus. The State Department of Public Health Laboratory utilizes the CDC’s priorities for testing that include:
- Hospitalized patients who have signs and symptoms compatible with COVID-19 in order to inform decisions related to infection control.
- Other symptomatic individuals such as, older adults and individuals with chronic medical conditions and/or an immunocompromised state that may put them at higher risk for poor outcomes (e.g., diabetes, heart disease, receiving immunosuppressive medications, chronic lung disease, chronic kidney disease).
- Any persons including healthcare personnel, who within 14 days of symptom onset had close contact with a suspect or laboratory-confirmed COVID-19 patient, or who have a history of travel from affected geographic areas within 14 days of their symptom onset.